 :max_bytes(150000):strip_icc()/PeriodicTablecolor-58b5d9105f9b586046df7d8a.jpg)
You’ll notice that metals occur to the left of the table, while non-metals fall on the right. .jpg)
Fortunately, most periodic tables use color to indicate whether the element is a metal, semi-metal, or non-metal. 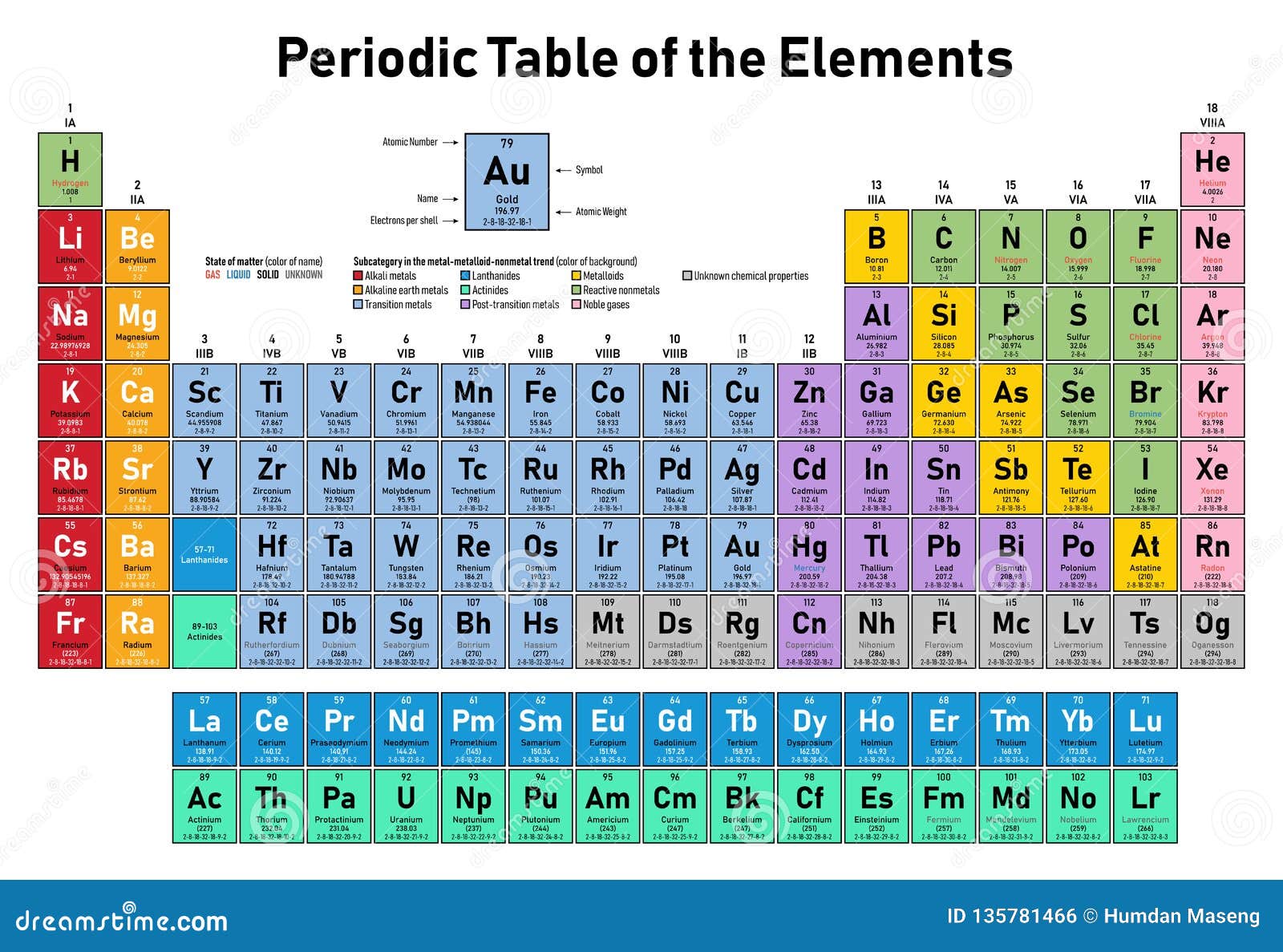
You can better understand the properties of an element by recognizing what type of element it is. The smallest bars often represent small amounts of more charged particles that you can ignore for identification purposes.Distinguish between metals, semi-metals, and non-metals. But most ions will have a charge of 1, and so you can ignore the division problem and just look at mass. 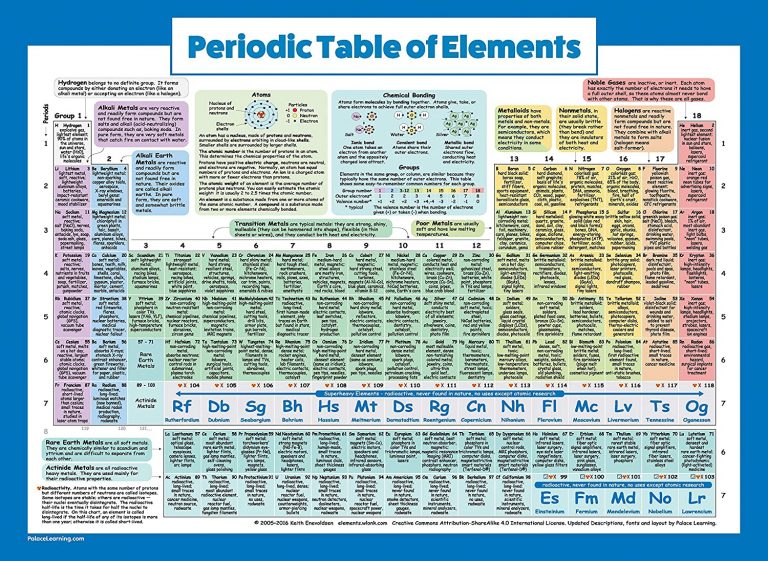
Technically, a mass spectrometer ionizes the sample and sorts by the ratio of mass to charge (or m/z).Adding these atoms together gives you the compounds HO (mass 1 + 16 = 17) and H 2O (mass 1 + 1 + 16 = 18). Only two of these match the atomic mass of an element: hydrogen (atomic mass 1) and oxygen (atomic mass 16). Let's say the tallest bar is at m/z 18, with short bars at 1, 16, and 17.Others (usually the larger ones) represent compounds, so that mass will equal the sum of masses of multiple atoms. Some values will match the atomic mass of an element that was part of the sample. To read the bar graph showing the results, check the "m/z" axis for the values of the taller bars. A mass spectrometer sorts the components of a sample by mass. Look for elements whose atomic masses match the graph. As you learn about different types of molecules, you'll learn to focus on just a few useful spots on the graph to save time. You can match the x-axis value at each peak to a table of known values to identify molecules. A more advanced spectroscope shows a detailed graph instead of a few lines.Different elements have different arrangements of electrons, which leads to different colors of bands. Want to know why this works? Electrons only absorb and emit light at very specific wavelengths (meaning specific colors).X Research source (Some types of spectra will show dark gaps instead of bright lines, but you can compare these the same way.) If your spectrum has all those same lines on it, the light came from the element lithium. For example, a lithium spectrum has a very bright, thick green line, and several other fainter ones in different colors.Each element releases a unique pattern of light, which you can see on the spectroscopy results, called "spectra". In spectroscopy, scientists examine how light interacts with an unknown material. Since this element has two electrons in 5p, count two elements into this row of the p-block to get the answer: tin.Ĭompare the spectra to the known spectra of elements. Go to the "p-block" on the right, and count rows down from 2p (boron) until you reach 5p (indium). For a more advanced example, the configuration 1 s 2 2 s 2 2 p 2.For example, if you are asked which element has 8 electrons, look for the element with atomic number 8: oxygen.All of the superscript numbers ( like this) are electron counts, so add all these together to find the total number of electrons. If you are a little further in your chemistry studies, you might be given an electron configuration to read. This number is the atomic number of the element, which you can look up on the periodic table. In a neutral atom, the number of electrons is exactly equal to the number of protons. The total electron count equals the atomic number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
